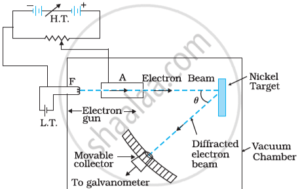
de Broglie Wavelength Equation :-
In 1924 Louis de Broglie proposed that just as light has both wave like and particle like properties, electrons also have wave like particles.
de Broglie proposed that the wavelength λ is associated with a particle of momentum p is given as
λ = h/p
Or

The equation is known as the de Broglie wavelength equation and wavelength λ of the matter wave is called de Broglie wavelength.
- For a photon, it moves as speed of light c Thus momentum of photon may be
p = mc = hν/c
since ν = c/λ
Therefore
p = hc/cλ = h/λ
λ = h/p
- Consider an electron (mass m, charge e) accelerated from rest through a potential V, the kinetic energy K of the electron equates the work done (eV) on it by the electric field –
K = eV
Now K = 1/2 mv² = p²/2m
p² = 2mK
p = √2meV
since λ = h/p
hence
λ = h/√2meV = h/√2mK
Substituting the numerical values of h, m and e we get
λ = 1·227/√V nm
Where V is the magnitude of accelerating potential in volts.
Numerical :-
- What is the de Broglie wavelength associated with (a) an electron moving with a speed of 5.4 X 10⁶ m/s and (b) a ball of mass 150 g travelling at 30 m/s ?
Solution.
(a) m = 9·1 x 10⁻³¹ Kg
v = 5·4 x 10⁶ m/s
λ = ?
p = mv
From de Broglie wavelength equation
λ = h/p = h/mv
= (6·63 x 10⁻³⁴ Js)/(9·1 x 10⁻³¹ Kg x 5·4 x 10⁶ ms⁻¹)
= (6·63 x 10⁻³⁴ Js)/(4·92 x 10⁻²⁴ Kgs⁻¹)
= 0·135 nm
(b) m = 150 g = 0·150 Kg
v = 30 m/s
λ = h/mv
= (6·63 x 10⁻³⁴ Js)/(9·1 x 10⁻³¹ Kg x 30 m/s)
λ = 6·63 x 10⁻³⁴ Js/4·50 Kgs⁻¹
= 1·45 x 10⁻³⁴ m