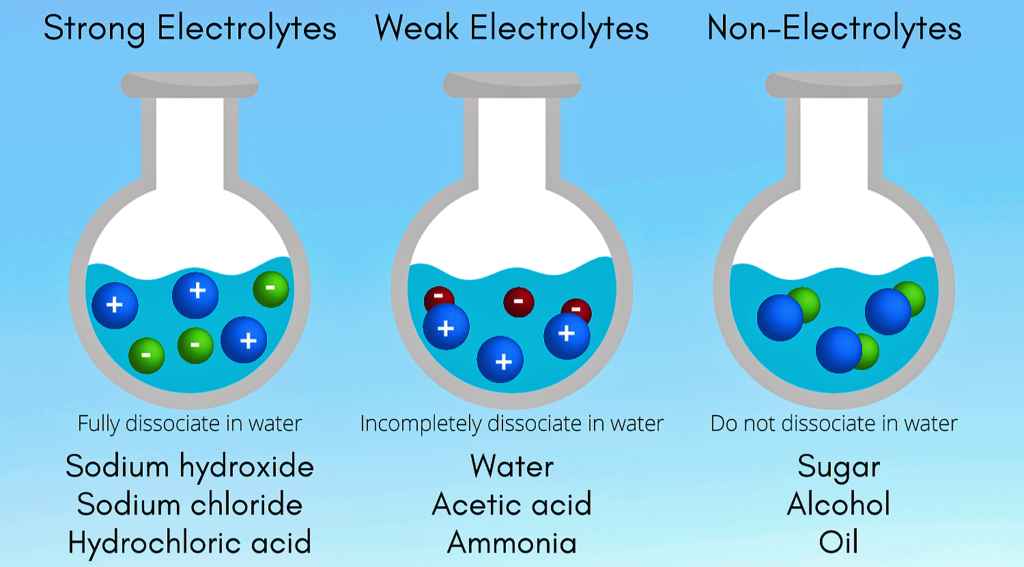
Electrolytes , Electolytic Cell and Electrochemical Cell
Electrolytes :- Electrolytes are substances that produce an electrically conducting solution when dissolved in a polar solvent , such as […]
Electrolytes , Electolytic Cell and Electrochemical Cell Read Post »