Nuclear Fission :-
Nuclear Fission is a reaction wherein a heavy nucleus is bombarded by neutrons and thus become unstable, which causes decompose or split into two nuclei with equivalent size and magnitude, with a great detachment of energy and the emission of two or three neutrons.
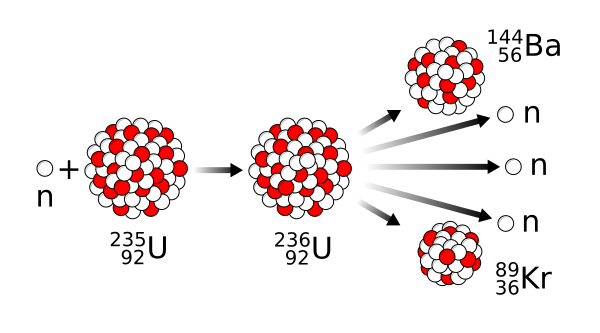
Example :- Isotope ₉₂U²³⁵ bombarded with a neutron breaks into two intermediate mass nuclear fragments.
₉₂U²³⁵ + ₀n¹ → ₉₂U²³⁶ → ₅₆Ba¹⁴⁴ + ₃₆Kr⁸⁹ + 3 ₀n¹
Another example :-
₀n¹ + ₉₂U²³⁵ →₉₂U²³⁶ → ₅₁Sb¹³³ + ₄₁Nb⁹⁹ + 4 ₀n¹
or
₀n¹ + ₉₂U²³⁵ → ₅₄Xe¹⁴⁰ + ₃₈Sr⁹⁴ + 2 ₀n¹
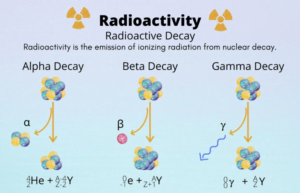
- The fragment products are radioactive nuclei. They emit particles in succession to achieve stable end product.
- The energy released in the fission reaction of nuclei like Uranium is of the order of 200 MeV per fissioning nucleus.
Let us
a nucleus with A = 240 breaking into two fragments each of A = 120
Then
Energy for A = 240 nucleus is about 7·6 MeV
Energy for two A = 120 fragments nuclei is about 8·5 MeV
Gaining in binding energy per nucleon is about 0.9 MeV
Here the total gaining binding energy = 240 × 0.9 = 216 MeV
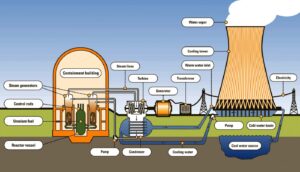
- The source of energy in nuclear reactors, which produce electricity, is nuclear fission.
- The enormous energy released in an atom bomb comes from uncontrolled nuclear fission.