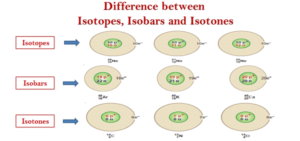
Mass Defect and Nuclear Binding Energy :-
The actual mass is always less than the sum of the individual masses of the constituent protons and neutrons because energy is removed when the nucleus is formed. This energy has mass, which is removed from the total mass of the original particles. This mass, known as the mass defect.
Mass defect can be calculated as the difference between observed atomic mass and that expected from the combined masses of its protons and neutrons.
i.e.

Nuclear Binding Energy:-
Nuclear binding energy is the energy required to split a nucleus of an atom into its component parts – protons and neutrons.
The binding energy of nuclei is always a positive number.
This energy required ‘E’ is related to the mass defect by
E = ∆mC²
Where ∆m = mass defect, C = velocity of light and E = nuclear binding energy.
- The energy equivalent of one atomic mass unit in MeV
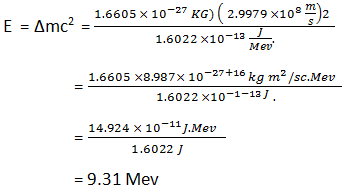
- If we separate a nucleus into its nucleons, we would have to supply a Total energy equal to binding energy ‘E’ to those particles.
- A more useful measure of the binding between the constituents of the nucleus is the binding energy per nucleon, which is the ratio of the binding energy ‘E’ of nucleus to the number of nucleons ‘A’
i.e. En = E/A
Nuclear Binding Energy Curve :-

Above figure is a plot of the binding energy per nucleon versus the mass number ‘A’ for a large number of nuclei.
The main features of the plot –
- The binding energy per nucleon is practically constant i.e. practically independent of the atomic number for nuclei of middle mass number ( 30 < A < 170 ).
2. The curve has a maximum of about 8.75 MeV for A = 238
Conclusion for these two observations –
(a) The force is attractive and sufficiently strong to produce binding energy of a few MeV per nucleon.
(b) The constancy of the binding energy in the range 30 < A < 170 is a consequence of the fact that the nuclear force is short ranged.
(c) A heavy nucleus say A = 240 has lower binding energy per nucleon compared to that of a nucleus with A = 120.
(d) Consider two very light nuclei ( A ≤ 10 ) joining to form a heavier nucleus. The binding energy per nucleon of the fused heavier nuclei is more than the binding energy per nucleon of the lighter nuclei.